Is Amazon failing you?Has Amazon kicked your product out?Amazon tested your product? FAILURE?We fix out of spec supplements
Click here for solutions you can trust!
Is Amazon failing you?Has Amazon kicked your product out?Amazon tested your product? FAILURE?We fix out of spec supplements
Click here for solutions you can trust!
Here's the Offer!
High-performance liquid chromatography (HPLC) is effective in determining the
How We can Help YOU Succeed in the Supplement Industry
Get Net 30 Terms on Latter 50%
Summary of 11 Tests
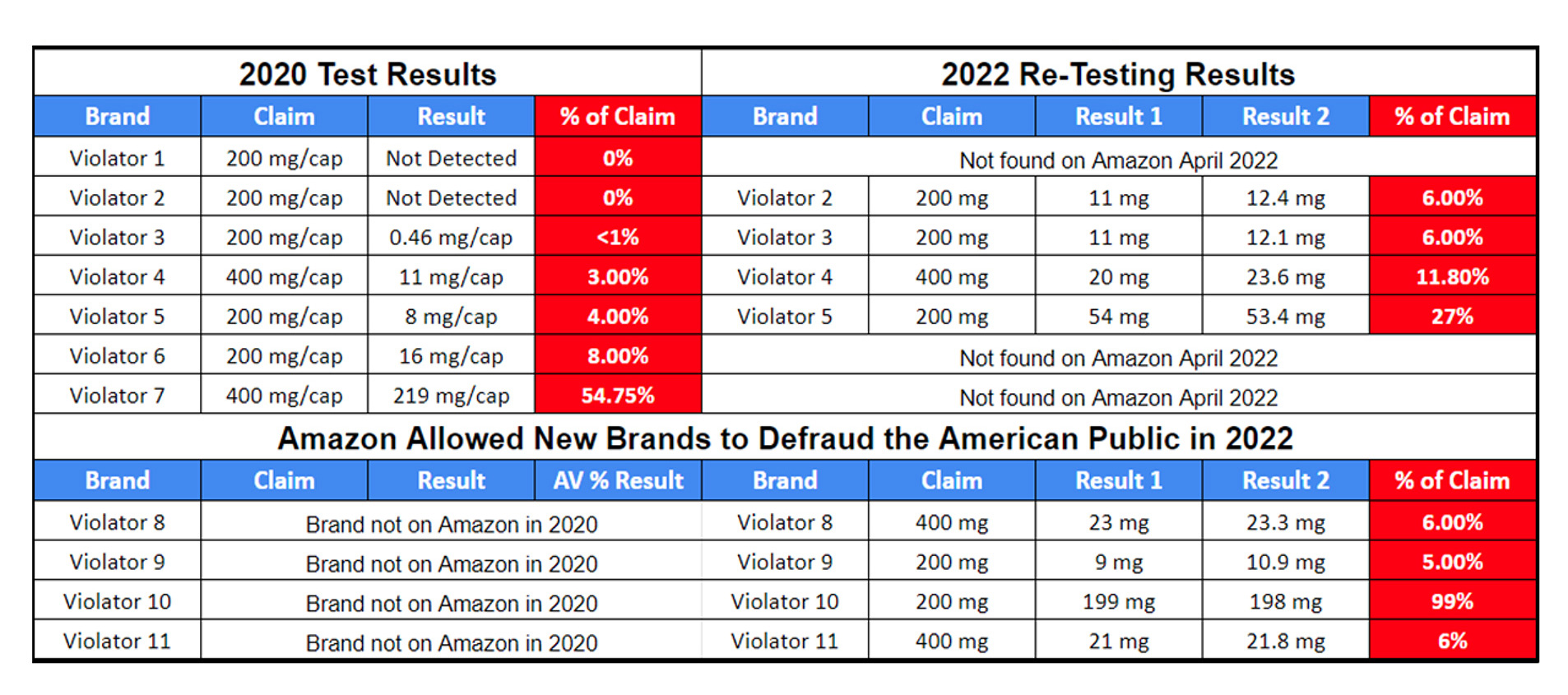
- No brand met label claims
- Two brands had no actives found
- One brand had less than 1%
- Most had 6% or LESS!

Why So Many Fake Supplements?
- Contract Manufacturers Lie to Brands that they are GMP Certified
NSF Needs to Improve!. - Factories and Brands Drylab and Falsify COAs.
NSF Needs to Improve!
- NSF certifications for Manufacturers, Warehouses and Distributors look the same.
- Resulting in factories holding a warehouse certification but falsely claiming manufacturing GMP certifications.
You need to check online…
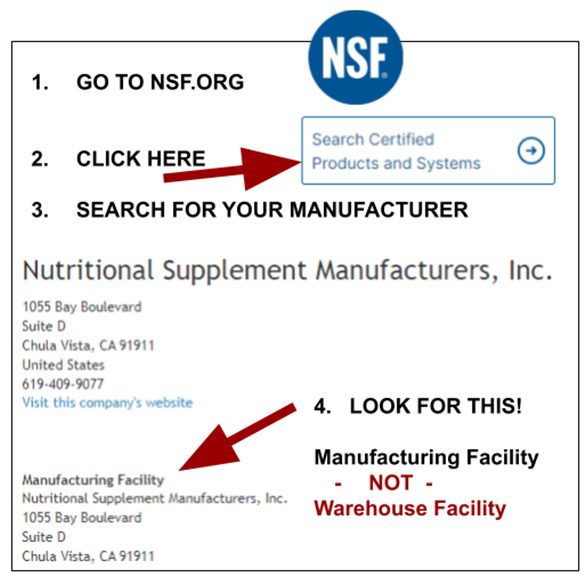
Don’t Get “Drylabbed”!
Amazon and other platforms:
Stop Taking PDFs at Face Value! Start Testing!
- COA is a fraud
- COA is altered
- Incorrect analytical methods
- Old COA
- Samples sent to lab not from lot listed
The term “dry lab” is one chemists use to describe a fraudulent experiment — one in which no actual chemistry is done (hence the word “dry”).

It takes time to change bad habits! Select manufacturers with GMP certifications for over 3 years. Call NSF at 1-734-773-4236 to check!
1000s OF SUPPLEMENTS ON AMAZON ARE SOON TO BE EXPOSED AS FAKE!
- THIS WILL INFLUENCE YOUR REPUTATION SIMPLY BECAUSE YOU SELL ON AMAZON (even if your supplement is to potency)
- NOW IS THE TIME TO HELP YOUR customers rest assured that your product is made in a GMP certified factory. That it has the potency stated on the label!

Stop Using Meaningless GMP Certification Icons!
Start Using Verified Icons!
Upgrade to Safer Tamper Evidence
Most Supplements Use Tamper Evidence Methods Invented in 1982


What We Focus On
Primary Output
- Encapsulations
- Tableting
- Liquids
Primary Competency
- Communicate Promptly to Clients
- Meet Label Claims
- Inhouse Lab
- Clean Labels
- Efficiency
- GMP Compliance
Innovations
- Nano / Liposomal
- Oil-2-Capsules With Banding
- Chewing Gum

Customers Desire Clean Labels
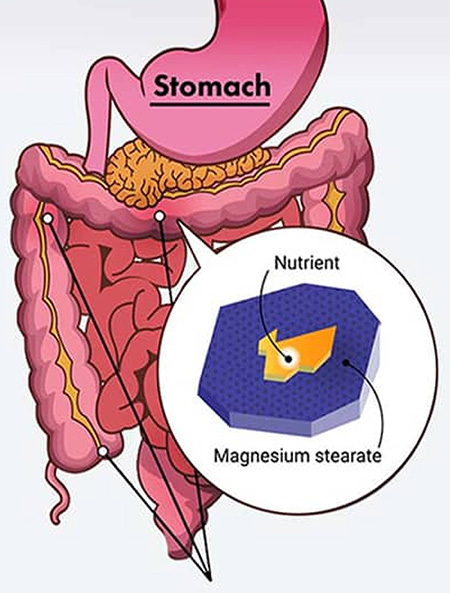
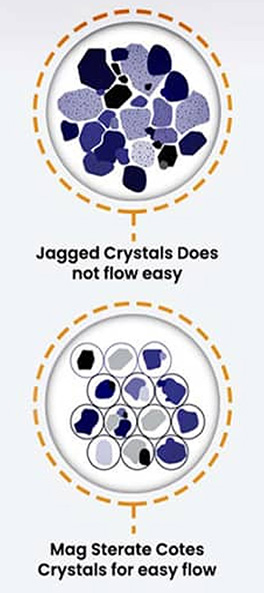

PureNSM manufactures both with and without magnesium stearates. The choice is yours. Manufacturing without mag stearate takes more labor hours and is therefore sometime more expensive. You need to decide what market you are going after.
Don’t Risk It!
- Always Request Being Listed on Your Contract Manufacturer’s Policy
- Don’t accept less than $5m

Quality Control and Quality Assurance
Quality Control (QC)
- Our inhouse lab is essential for manufacturing quality products with correct overages to meet potency claims
Quality Assurance (QA)
- Our quality assurance team is the nervous system of our operations. They review production records, maintain Facility and equipment and more
Lab Equipment/Methods
- HPTLC
- ICP-MS
- FTIR
- Microbiology Testing
- Rapid Micro – Solaris
- Micro Plating Methods
- 3M Micro Testing
- BSL-2 Laboratory
- HPLC
- Tablet Testing




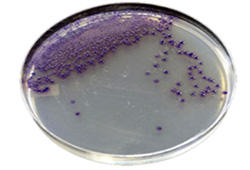
HPTLC - High Performance Thin Layer Chromatography
- About 30% of all botanicals are adulterated (wrong species, wrong plant part, extracted plant material leftover)
- The only way to ID the plant material is with HPTLC.
- HPTLC is better than DNA analysis, because of DNA damage in plant extract and powders.
- HPTLC is not to be confused with FTIR or HPLC
- HPTLC is a systematic whet chemistry
- We have over 350 methods developed
- FTIR
- If we don’t have the methods we work with Alchemist Labs
- You WILL have correct plant parts in your product. You WILL meet potency claims.

Compostable Bottles
- Our inhouse lab is essential for manufacturing quality products with correct overages to meet potency claims.

Huge Production Capacity Available
- Huge Production Volume! 91 million caps per month! Highly trained line-workers


GMP Audited Warehouses
- Humidity and Temperature Controlled
- Raw Materials and Finished Goods
- Your Products Are Safe With Us!


GMP Audited Pick and Pack Warehouse
- Ship your finished goods with us!
- We connect to your website and ship without delay





Nano/Liposomal
HIGH PRESSURE HOMOGENIZER
- We offer to process your emulsions on our high-pressure homogenizer from IDEX, model M815.
-
Flow rates to 1.0-1.2 Liter/min
Pressure to 2068 bar (30,000 psi) - Our Chemical Engineer can make any liquid supplement more absorbable. Particle size can go to below 100nm
Ideal for:
- Expensive ingredients
- Fat soluble (hydrophobic) ingredients
- Create formulas for people with poor digestion

- With our excellent formulator we can reduce particle size of any nutraceutical.
- We can then have them tested on a particle size analyzer.
Questions
What do you need a quote for?
- Existing brand? Please answer questions in group 1.
- New brand? Please answer questions in group 2.
Questions: Group 1
For Existing Brands
- Are you switching from another manufacturer altogether or adding a backup manufacturer?
- Has this exact formula been manufactured with prior manufacturers?
- Are you aware if the manufacturer had any problems with getting the projects done in the past?
- What did you not like about your current or previous manufacturer?
- What did you like?
- When do you need the products?
- Where do you sell your products?
- Do you have the production records? (important for tablets and flavored liquids)
- Do you want to supply any of the raw materials?
- Do you have a preferred raw material vendor?
- Are you exporting this?
- Estimated quantity needed for the reminder of this year?
- Estimated quantity needed over next calendar year for all your SKUs?
- Is it important that we don’t use magnesium stearate or other processing aids?
Questions: Group 2
For New Brands
- Are you looking for stock products or your own unique formula?
- If custom, do you have a formula, or do you need formulation assistance?
- When do you need the products?
- How many?
- Where do you plan to sell your products?
- How much experience do you have in this industry?
- Are you an influencer? How many followers do you have?
- Do you plan to export?
- Do you need help with marketing?
- Do you have a budget for this project?
- Is it important that we don’t use magnesium stearate or other processing aids?

