CERTIFICATIONS
PlureNSM has two GMP Certifications as Supplement Manufacturer. Both can be verified instantly, as follows. Never rely on PDFs of GMP certifications.
- You can verify our UL certification HERE
- Scroll down
- Click Plus (+) sign next to “Retail Certification Program (RCP)”
- Search PureNSM
- You can verify our NSF certification HERE
- Go to www.nsf.org
- Look for this tab on lower left”Search Certified Products and Systems”
- It is next to “Search NSF.org”
- A search field will appear
- Now search “PureNSM”
- You can verify our UL certification HERE
- You can verify our NPA certification HERE.
- On your keyboard type Ctrl + F
- Search PureNSM
Warning: While our certifications are real and easily verifiable, other contract manufacturers have been known to provide fake certifications. If you get an NSF or UL certification you need to verify if they are current and if they are issued to the entity you are wanting to have manufacture for you. Do so periodically.
All our products come with a $5,000,000 product liability insurance
We provide Certificate of Liability Insurance

Two main reasons CMOs don’t follow GMP regulations
Competing on cost is one but there is more to it. The second and more important reason is that most CMOs don’t realize that suppliers of raw materials for supplements are governed by a different set of regulations than manufacturers are.
Regulations governing raw material vendors for supplements are subject to a less stringent regulation called 21 CFR 117 and not 21 CFR 111, which is the better-known regulation governing supplement manufacturers.
FDA claims raw materials vendors don’t need to test as rigorously as supplement manufacturers because they also sell to the fragrance and cosmetics industries. This makes it all the more important for brand owners to select quality manufacturers that follow strict quality control protocols.
Unfortunately, many brands seeking to hire supplement manufacturers don’t realize this and neither do the factories they hire to make their products. This is why it’s so frustrating that NSF has made it even more confusing and difficult to gauge if the GMP certification from your manufacturer can be trusted. See more here, Don’t rely on PDFs of GMP certifications from NSF!
It’s important for brand owners to manufacture with CMOs that own and operate their own laboratory on location. Most importantly it’s important that they ensure that all lab equipment is calibrated to work as desired.
Perhaps the most important routine test performed in an inhouse laboratory owned and operated by manufacturers is microbiology analysis. Did you know that such equipment can’t be verified to work as desired if the micro lab is not equipped to handle live Salmonella and E. Coli? Find out here: Preventing Disaster with Microbiology Calibration. Taking microbiology testing seriously is very important. Not so long ago people got blinded by artificial tears. It can happen in the supplement industry, FDA recalled 859 brands manufactured by one factory.
Don’t rely on PDFs of GMP certifications from NSF!
We don’t send PDFs of GMP certifications anymore because we don’t want our customers to get into the habit of relying on them in general. If we send PDFs, our customers may rely on PDFs from our competitors which in many cases are misleading.
GMP certifications from NSF don’t tell the whole story. This is because NSF issues the same PDF document for 3 different types of companies:
- GMP certifications for a Distribution Facility
- GMP certifications for a Warehouse Facility
- GMP certifications for a Manufacturing Facility
The ONLY way to see the difference is to go online and look above the 2nd listing of the company name (see directions on image attached and below). There it’s stated if the certification is for one of the above three types of companies.
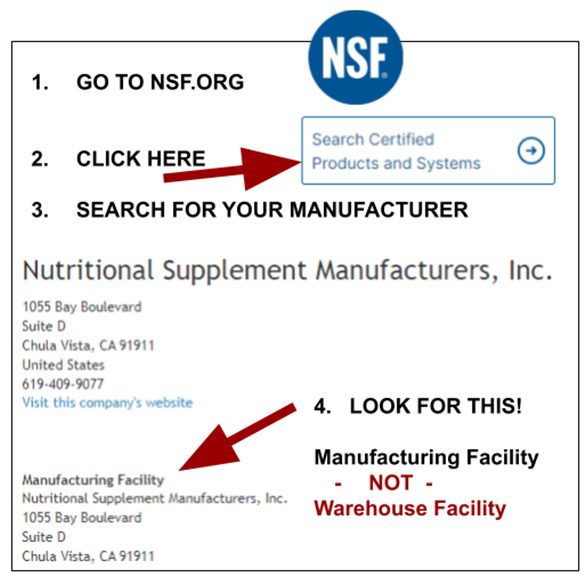
Of course, you want your CMO to hold a GMP certification for a Manufacturing Facility.
The fact that NSF does not put this on their PDFs has caused lots of confusion and has caused many manufacturers to only hold a GMP certification for a Warehouse Facility and then claim Manufacturing certification.
This is of course something we at PureNSM don’t like and it’s therefore we have stopped sending out PDFs and have chosen not to renew our NSF certification.
Preventing Disaster with Microbiology Calibration
All analytical equipment needs to be calibrated. That goes without saying. However what people are overlooking in the supplement industry is the verification (calibration) that the microbiology equipment used to detect Salmonella and E. Coli actually works.
Although such pathogens are a required test, most inhouse laboratories with manufacturers don’t perform any validation on their microbiology equipment for pathogens.
This is because in order to perform such validation you need to handle live culture of pathogens. In order to handle live culture you need to take extra precautions. In other words you need a BSL-2 Microbiology Laboratory. Such labs are designed with specific safety measures to protect both the laboratory personnel and the environment.
PureNSM operates a BSL-2 Microbiology Laboratory. With us you can rest assured that you will not need to recall your supplements.
Our Business License

PureNSM Has Experience With Certifications













